Phospholipids:
Phospholipids are a unique group of lipids. In a phospholipid, two fatty acid chains are bound to a phosphate group. The phosphate group contains lots of oxygen and is highly polar, and so it is hydrophilic. But the fatty acid chains are nonpolar and so hydrophobic. This gives phospholipids a sort of split personality, because the phosphate-containing heads are strongly attracted to water, whereas the fatty-acid tails are not. Molecules such as phospholipids that contain both polar and nonpolar portions are said to be
amphipathic.
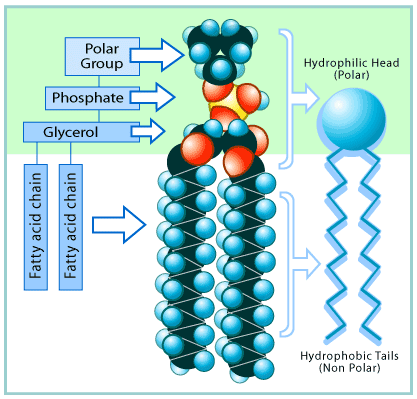
The structure of a phospholipid molecule
Because the hydrophilic heads of phospholipids are attracted to water and the hydrophobic tails repelled by it, phospholipids will spontaneously arrange themselves into
bilayers in water. A phospholipid bilayer consists of two layers of phospholipid molecules arranged so that the water-loving heads point outward and toward the surrounding water, while the tails point inward, away from the water.
Phospholipids are the primary components of the
cellular membranes that surround and hold together all cells. Additionally, most of the
organelles within a cell are either made of or surrounded by membranes that are mostly made of phospholipids.